Services
Center for Advanced Medicine and Clinical Research
Advanced Medicine Division
Mission
To explore and advance potential new treatments from laboratory discoveries.
- Research Support
Clinical Sequencing
Mission
To support appropriate medical care and clinical studies through genome analysis using next-generation sequencers.
Cell Banking
Mission
To construct bioresource banks capable of providing high-quality clinical specimens for use in omics-based research and others.
- Material Preparation
Material Preparation
Mission
To prepare material for gene medicine, material for cell medicine, and material for regenerative medicine by utilizing biomaterial preparation equipment.
- Information Technology
Mission
To comprehensively operate the CAMCR-related systems.
- CAMCR IP Strategy
Mission
To efficaciously manage and operate intellectual properties.
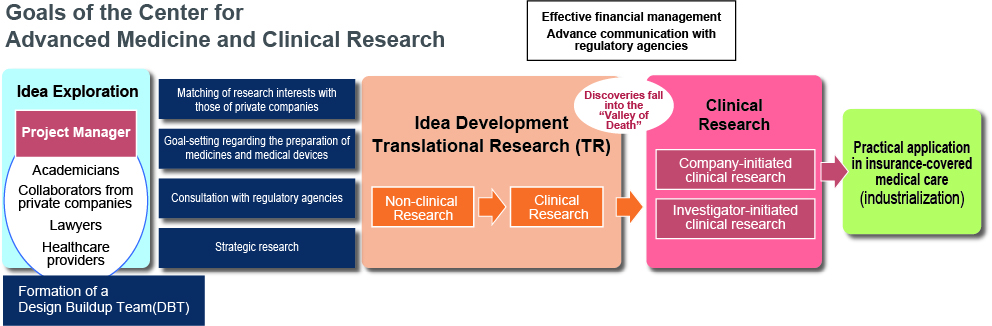
Clinical Research Division
Mission
To deliver the findings in high-standard clinical research from Nagoya and Chubu to the world.
- Planning
Project Planning and Management / Medical Writing
Mission
To accelerate and streamline clinical studies.
Regulatory
Mission
To forward in academia the rapid development of more efficacious, safer, and innovative medical technologies (e.g., drugs, medical devices, and regenerative medicine products) based on regulations, notifications, evidence, and others.
- Management
Coordination
Mission
To provide and conduct CRC services for allowing the smooth conduct of clinical studies and clinical trials.
Patient Support Services
Mission
To afford patient services for allowing the smooth conduct of clinical studies and clinical trials in an attempt to improve patient satisfaction.
Multi-center/International Joint Clinical Trial Support
Mission
We will promote multicenter and international clinical trials.
- Unapproved Medication and Others Management
Mission
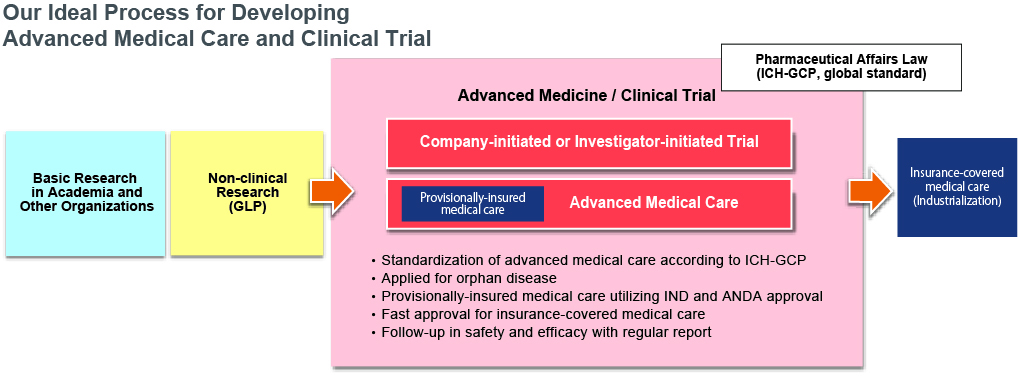
To support advanced medical care by accelerating high-quality clinical research.
Data Coordinating Center
Data Science Division
Mission
Implementing data science solutions for supporting medical research, and accelerating advances in public health and medicine.
- Statistics Analysis
Mission
To enhance quality of medical research conducted in central Japan through excellent use of methods in biostatistics and bioinformatics.
Clinical Data Quality Control Division
Mission
To control the quality and integrity of clinical trial data by clinical monitoring and data management.
- Clinical Data Quality Control
Monitoring
Mission
To forward in academia the conduct of highly confident clinical studies based on regulations, notifications, protocols, and others, to protect the human rights, maintain the safety, and improve the welfare of subjects, as well as to contribute to the reliability of clinical study results.
- Clinical Network Management
System Administration
Mission
To forward patient enrollment in investigator-initiated clinical trials and clinical studies through subject recruiting using the patient accumulation registry and to support the clinical network collaboration inside and outside the Chubu region.
- Clinical Data Management
Data Management
Mission
To appropriately design scientific, rational, and ethical clinical research. To support investigators in an attempt to guarantee the reliability thereof and from objective and professional standpoints.
Quality Assurance
Mission
To assure that the clinical study was conducted and its data were prepared, recorded, and reported in compliance with regulations, guidelines, and others and in accordance with the approved protocol.
